

Each of the corner atoms is the corner of another cube so the corner atoms are shared among eight unit cells. The face centered cubic structure has atoms located at each of the corners and the centers of all the cubic faces (left image below). These hardening mechanisms will be discussed latter. It should be noted that there are other important mechanisms for hardening materials, such as introducing impurities or defects which make slipping more difficult. When the metal is deformed, the planes of atoms must slip over each other, and this is more difficult in the bcc structure.
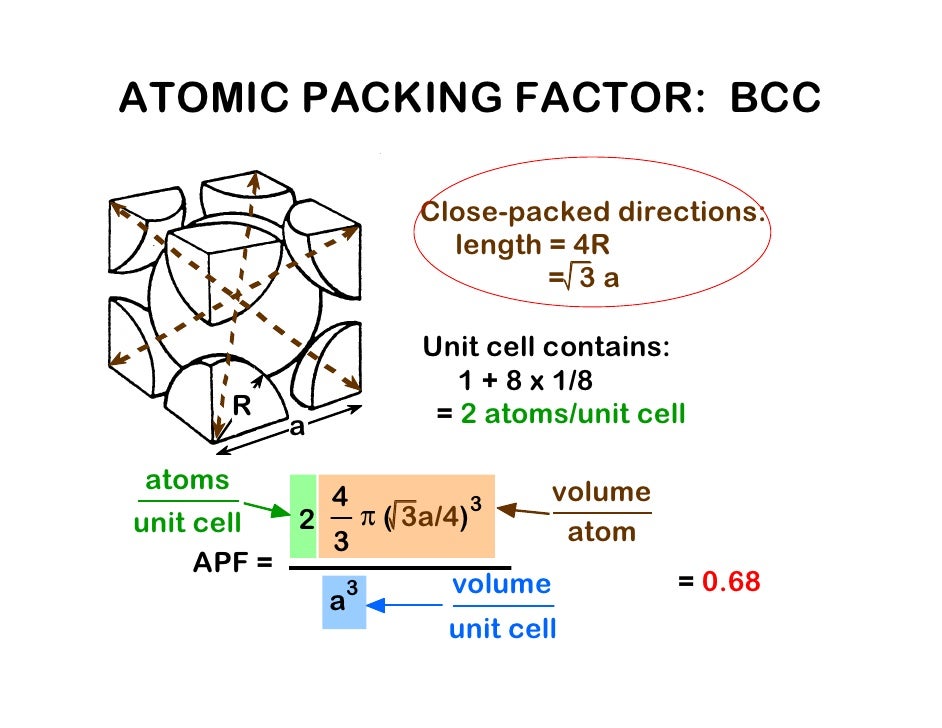
Metals which have a bcc structure are usually harder and less malleable than close-packed metals such as gold. Some of the materials that have a bcc structure include lithium, sodium, potassium, chromium, barium, vanadium, alpha-iron and tungsten. The bcc unit cell has a packing factor of 0.68.
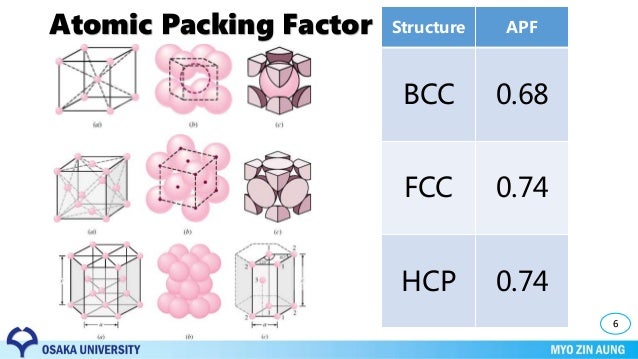
The volume of atoms in a cell per the total volume of a cell is called the packing factor. The bcc structure is often the high temperature form of metals that are close-packed at lower temperatures. The bcc arrangement does not allow the atoms to pack together as closely as the fcc or hcp arrangements.
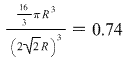
The image below highlights a unit cell in a larger section of the lattice. The bcc unit cell consists of a net total of two atoms one in the center and eight eighths from corners atoms as shown in the middle image below (middle image below). It is said to have a coordination number of 8. The body-centered cubic unit cell has atoms at each of the eight corners of a cube (like the cubic unit cell) plus one atom in the center of the cube (left image below). Since these structures are most common, they will be discussed in more detail. However most metals and many other solids have unit cell structures described as body center cubic (bcc), face centered cubic (fcc) or Hexagonal Close Packed (hcp). Physics of Nondestructive Evaluation > Materials and Processes > Primary Metallic Crystalline Structures Primary Metallic Crystalline StructuresĪs pointed out on the previous page, there are 14 different types of crystal unit cell structures or lattices are found in nature.

 RSS Feed
RSS Feed
